VALNEVA SE WKN: A0MVJZ ISIN: FR0004056851 Forum: Aktien Thema: Hauptdiskussion
2,552
EUR
-7,54 % -0,208
17:57:14 Uhr,
Lang & Schwarz
Kommentare 303.709
Henry1966,
18.04.2025 10:28 Uhr
0
Weiter:
To date, Valneva has supplied approximately 80,000 doses of IXCHIQ(®) in the
United States, Canada and Europe. No further SAEs have been reported since
January 2025 globally, and Valneva has not identified any safety signal concerns
that are inconsistent with the U.S. product label through its ongoing post-
marketing safety monitoring, including its periodic safety reports and routine
signal detection activities, which are shared with the U.S. Food and Drug
Administration (FDA).
Juan Carlos Jaramillo, M.D., Chief Medical Officer of Valneva, commented,
'Valneva is committed to the highest standards of safety, and the safety profile
of IXCHIQ(®) remains unchanged and positive. We respect the ACIP recommendation
and agree on the importance of continuing the stringent safety surveillance
protocols that are in place. We encourage providers to assess the benefit/risk
of vaccination based on the individual's medical history and upcoming travel, in
line with the current recommendation.'
The recommendation from ACIP is pending final approval by the Director of the
CDC and the U.S. Department of Health and Human Services.
About Chikungunya
Chikungunya virus (CHIKV) is a mosquito-borne viral disease spread by the bites
of infected Aedes mosquitoes which causes fever, severe joint and muscle pain,
headache, nausea, fatigue and rash. Joint pain is often debilitating and can
persist for weeks to years(1).
In 2004, the disease began to spread quickly, causing large-scale outbreaks
around the world. Since the re-emergence of the virus, CHIKV has now been
identified in over 110 countries in Asia, Africa, Europe and the Americas(2).
Between 2013 and 2023, more than 3.7 million cases were reported in the
Americas(3) and the economic impact is considered to be significant. The medical
and economic burden is expected to grow with climate change as the mosquito
vectors that transmit the disease continue to spread geographically. As such,
the World Health Organization (WHO) has highlighted chikungunya as a major
public health problem.(4)
Henry1966,
18.04.2025 10:21 Uhr
0
Auf dütsch
18. April (Reuters) – Valneva SE:
* VALNEVA AKTUALISIERT DIE ACIP-EMPFEHLUNG FÜR SEINEN
CHIKUNGUNYA-IMPFSTOFF IXCHIQ® FÜR US-REISENDE
* VALNEVA SE – CDC AKTUALISIERT DIE EMPFEHLUNG FÜR VALNEVAS
CHIKUNGUNYA-IMPFSTOFF
* VALNEVA SE – ACIP BEHÄLT DIE IXCHIQ-EMPFEHLUNG FÜR REISENDE IN AUSBRUCHSGEBIETE BEI
((Reuters.Briefs@thomsonreuters.com;))
Henry1966,
18.04.2025 10:20 Uhr
0
April 18 (Reuters) - Valneva SE :
* VALNEVA PROVIDES UPDATE ON ACIP RECOMMENDATION FOR ITS
CHIKUNGUNYA VACCINE IXCHIQ® AMONG U.S. TRAVELERS
* VALNEVA SE - CDC UPDATES RECOMMENDATION FOR VALNEVA'S
CHIKUNGUNYA VACCINE
* VALNEVA SE - ACIP MAINTAINS RECOMMENDATION FOR IXCHIQ FOR
TRAVELERS TO OUTBREAK AREAS
((Reuters.Briefs@thomsonreuters.com;))
Henry1966,
18.04.2025 10:17 Uhr
0
Saint Herblain (France), April 18, 2025 - Valneva SE
(https://valneva.com/?lang=fr) (Nasdaq: VALN; Euronext Paris: VLA), a specialty
vaccine company, today announced that during its regular meeting on April 16,
the U.S. Centers for Disease Control and Prevention's (CDC) Advisory Committee
on Immunization Practices (ACIP) updated its recommendation for use of Valneva's
single-dose chikungunya vaccine IXCHIQ(®) for the prevention of disease caused
by the chikungunya virus (CHIKV). ACIP maintained its current recommendation for
IXCHIQ(®) for persons aged >=18 years traveling to a country or territory where
there is a chikungunya outbreak. Additionally, it may be considered for persons
aged >=18 years traveling or taking up residence in a country or territory
without an outbreak but with elevated risk for U.S. travelers if planning travel
for an extended period of time e.g., six months or more.
The ACIP also voted to recommend a precaution related to the use of IXCHIQ(®) in
persons aged >=65 years. This precaution is a response to an ongoing
investigation by the CDC of six cases of serious adverse events (SAEs),
including five hospitalizations, among persons aged 67-86 years after
vaccination with IXCHIQ(®). These SAEs were reported through the Vaccine Adverse
Event Reporting System (VAERS), which is intended to be an early warning system
to identify potential safety issues but generally cannot determine if adverse
events are caused by a vaccine.
All of the individuals who were hospitalized had pre-existing comorbidities, and
ACIP concluded that while plausible, no causal association with IXCHIQ(®) could
be determined for all cases and that further investigation is warranted.
ACIP also noted that for individuals aged >=65 years, vaccination with IXCHIQ(®
)might be indicated in certain higher-risk settings (e.g., outbreak), given the
known risks for severe chikungunya disease and hospitalization in this age
group. For example, earlier this month Valneva supplied 40,000 doses of
IXCHIQ(®) to France's Island of La Réunion where the local public health agency,
the Agence Régionale de Santé (ARS), is prioritizing vaccination of adults aged
65 and over, especially those with comorbidities, to protect residents during
the ongoing chikungunya outbreak. Valneva recently received confirmation from
ARS for an order of 50,000 additional doses of IXCHIQ(®) as part of its ongoing
effort to manage this outbreak.
s
speedbeiryanair,
18.04.2025 9:08 Uhr
0
Richtig, Valneva ist eine wahre Perle! Von dieser Unternehmensführung können unsere Politiker in Deutschland noch was lernen! 🤣
Ranisch69,
18.04.2025 8:39 Uhr
0
Frohe Ostereiersuche 😀
DividendenGraf,
18.04.2025 6:28 Uhr
0
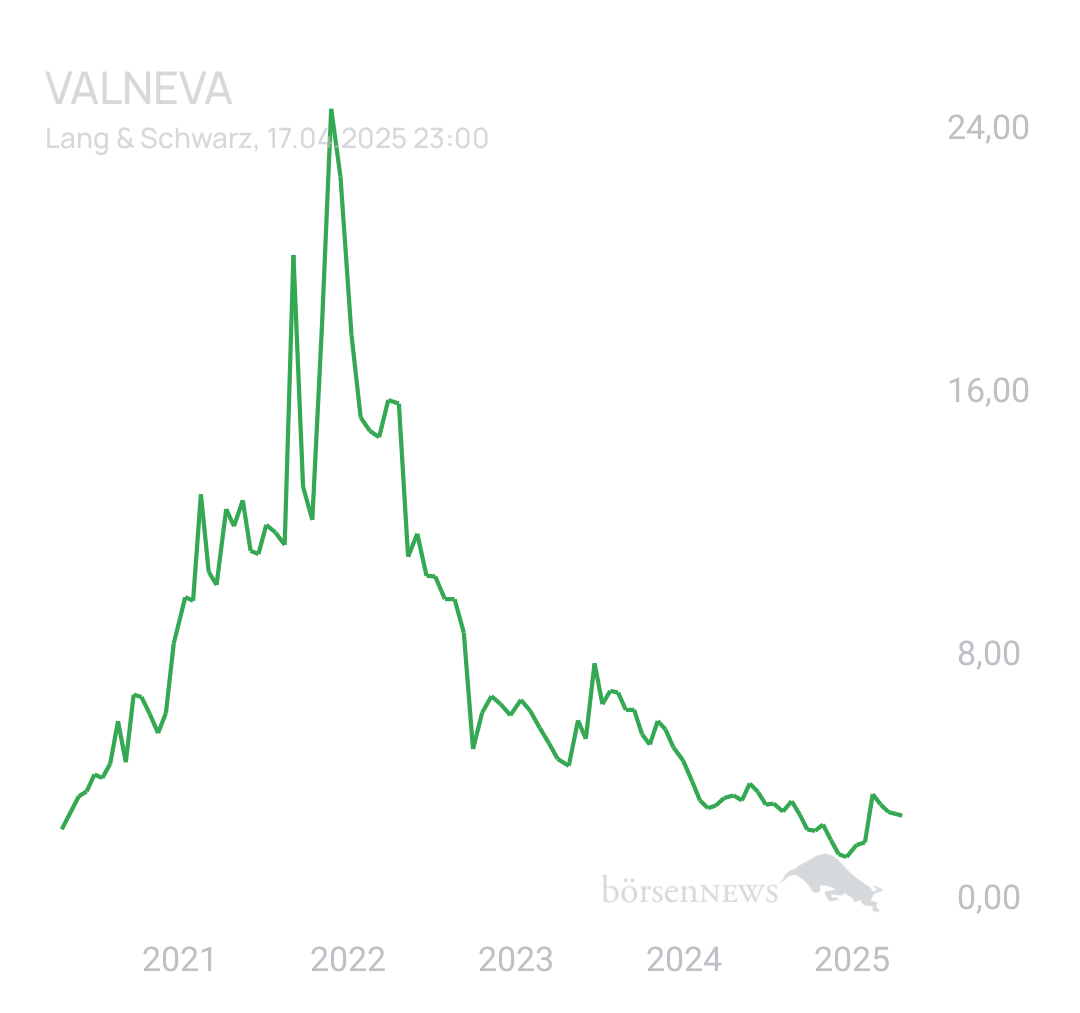
Wenn man vor 5 Jahren eingestiegen wäre, wäre man jetzt schon 15% im Plus. D.h. im Durchschnitt macht Valneva eine Performance von 3% pro Jahr. Was für eine Performance und das bei dem Risiko, einfach stark 💪 💪 💪 💪
B
Boersenhan,
17.04.2025 21:36 Uhr
5
Zielgruppe wird seit heute vergrößert.
https://www.linfo.re/la-reunion/societe/depuis-ce-jeudi-la-vaccination-gratuite-contre-le-chikungunya-est-elargie-18-a-64-avec-des-comorbidites-et-65-ans-et-plus
Win20002,
17.04.2025 21:27 Uhr
0
Danke, das wünsche ich auch. Frohe Ostern 🐣🐇
Jens08,
17.04.2025 18:40 Uhr
0
95🚀
KaRoLa1960!,
17.04.2025 16:16 Uhr
4
Ich wünsche Euch schöne dicke, bunte Eier!
Frohe Ostern 👋
KaRoLa1960!,
17.04.2025 16:12 Uhr
0
🤦
s
speedbeiryanair,
17.04.2025 13:42 Uhr
0
Hallo Nixblicker, Maxitaurus und Lapilli59
m
maxitaurus,
17.04.2025 13:16 Uhr
0
Hallo Nixblicker
Mehr zu diesem Wert
|
|
Thema | ||
|---|---|---|---|
| 1 | VALNEVA SE Hauptdiskussion | ||
| 2 | Valneva ohne Politik | ||
| 3 | Wobsy Valneva | ||
| 4 | Valneva Insolvenz möglich? | ||
| 5 | Valneva direkt | ||
| 6 | Valneva für EK über 15 | ||
| 7 | Fallneva | ||
| 8 | Valneva Totimpfstoff | ||
| 9 | Valneva - Tenbagger | ||
| 10 | Daytrade Valneva |
Meistdiskutiert
|
|
Thema | ||
|---|---|---|---|
| 1 | INTEL Hauptdiskussion | -9,14 % | |
| 2 | RHEINMETALL Hauptdiskussion | -1,84 % | |
| 3 | Dax Prognose | ±0,00 % | |
| 4 | New Highland Critical Minerals Registered (A) Hauptdiskussion | -46,23 % | |
| 5 | DAX Hauptdiskussion | ±0,00 % | |
| 6 | NVIDIA Hauptdiskussion | -0,93 % | |
| 7 | Investmentchancen | -4,76 % | |
| 8 | Security der nächsten Generation | -8,09 % | |
| 9 | Das neue Dax Prognose Forum | ±0,00 % | |
| 10 | ATOS Hauptdiskussion | -1,26 % | Alle Diskussionen |
Aktien
|
|
Thema | ||
|---|---|---|---|
| 1 | INTEL Hauptdiskussion | -8,94 % | |
| 2 | RHEINMETALL Hauptdiskussion | -1,96 % | |
| 3 | New Highland Critical Minerals Registered (A) Hauptdiskussion | -46,23 % | |
| 4 | NVIDIA Hauptdiskussion | -1,09 % | |
| 5 | Investmentchancen | -5,54 % | |
| 6 | Security der nächsten Generation | -9,37 % | |
| 7 | Palantir ohne Schnickschnack | -1,51 % | |
| 8 | ATOS Hauptdiskussion | -1,26 % | |
| 9 | Beyond Meat Hauptdiskussion | -3,88 % | |
| 10 | Novo Nordisk nach Split | +0,53 % | Alle Diskussionen |